Draw The Lewis Structure For Ibr
9.2: Interpreting Lewis Structures
-
- Last updated
- Save as PDF
- Page ID
- 170029
Learning Objectives
- To identify bonding and nonbonding electron pairs within a Lewis Structure.
- To identify bond order for bonds within a Lewis Structure.
- Understand the relationship between bond order, bond distance, and bond energy.
- To use electronegativities to determine bond polarity.
- To assign formal charges to each atom in a Lewis Structure.
This text assumes previous knowledge of Lewis Dot Structures. The following section will review how to draw structures, but first we will address some key Lewis Dot Structure ideas.
Interpreting Lewis Structures
A Lewis structure contains symbols for the elements in a molecule, connected by lines and surrounded by pairs of dots. For example, here is the Lewis structure for water, H2O.
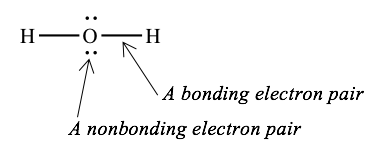
Each symbol represents the nucleus and the core electrons of the atom. Here, each "H" represents the nucleus of a hydrogen atom, and "O" represent the nucleus and the two core electrons of the oxygen atom. The dots represent nonbonding valence electrons. There are four nonbonding valence electrons on the oxygen atom. Each line represents a pair of bonding electrons, which is shared between two atoms. This is typically called a single bond.
When there are two lines connecting a pair of atoms, there are four bonding electrons (two pairs) between the atoms. This is called a double bond.
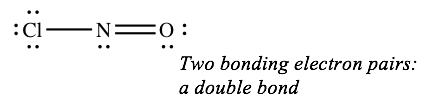
Three lines between a pair of atoms means six bonding electrons (three pairs), and is called a triple bond.

Bond Order, Bond Distance, and Bond Energy
The number of electron pairs in a bond is called the bond order.
C–C single bond bond order = 1
C=C double bond bond order = 2
C\(\equiv\)C triple bond bond order = 3
The bond order is directly related to the length and strength of a bond.
Higher bond order = stronger bond (higher bond energy)
Higher bond order = shorter bond (smaller bond distance)
Here is an example, comparing the lengths and strengths of bonds between carbon and nitrogen.
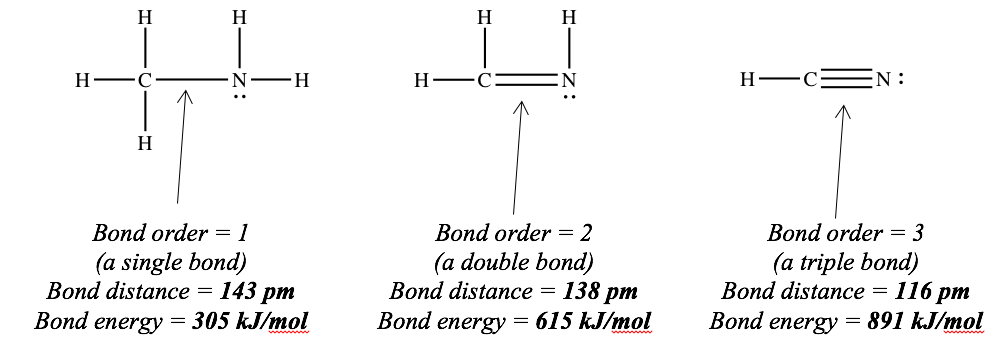
Warning : this only works when you compare bonds between the same pair of elements. You can safely say that a C=N double bond is shorter than a C–N single bond, but you cannot safely say that a C=N double bond is shorter than a C–H single bond. (In fact, the C–H bond is shorter!)
The bond energy and bond distance depend on the bond order, but they normally do not depend very much on the other atoms and bonds in the molecule. For example, the circled N–H bonds in the following molecules are roughly the same length.
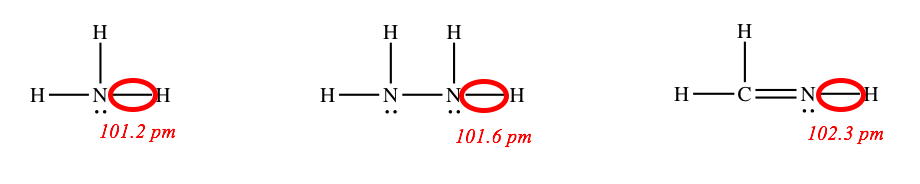
Bond Polarity
Whenever a covalent bond links atoms of different elements, the bond will be polar. A polar bond is one in which the atoms have unequal electrical charges. Generally, one atom will be negatively charged and the other will be positively charged. For example, the bond in HCl is polar. The hydrogen atom is positively charged and the chlorine is negatively charged. We can represent this as follows:
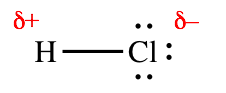
The symbol \(\delta\) means "a little bit." We use it to show that the atoms are not ions with integer charges (+1 and –1); the hydrogen atom is slightly positive and the chlorine atom is slightly negative.
A bond is polar when the two atoms have different attractions for electrons. In the case of HCl, chlorine attracts electrons more strongly than hydrogen does. Therefore, the two bonding electrons move toward the chlorine atom, making it negatively charged (and the hydrogen positively charged).
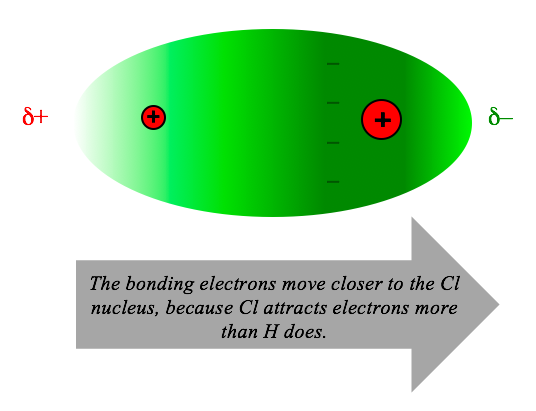
Electronegativity: determining which atom is positive and which is negative.
To figure out which atom is positively charged and which is negatively charged in a covalent bond, we use the electronegativities of the atoms. Electronegativity is a number that measures how strongly an atom attracts electrons, both its own electrons and those of other atoms. Here is a table that shows the electronegativities for the representative elements.
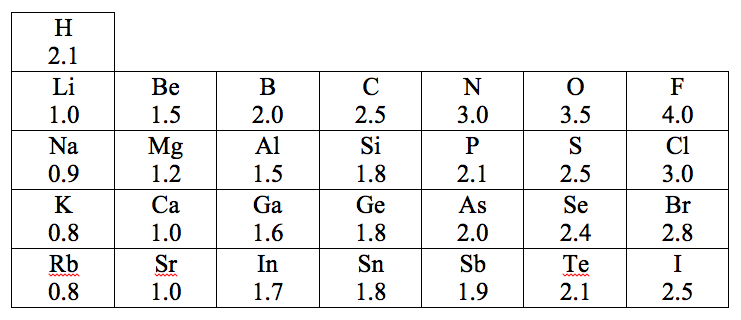
The elements with the lowest electronegativities are on the far left side of the table. These are elements that have weak attractions for electrons; they do not attract electrons from other atoms, and they do not hold their own valence electron(s) very tightly. These atoms tend to be positively charged when they form compounds. The elements with the highest electronegativites are in the upper right corner of the table. These are elements that have strong attractions for electrons; they can "steal" electrons from other atoms, and they hold their own valence electrons very tightly. These atoms tend to be negatively charged when they form compounds. Note that hydrogen has an intermediate electronegativity; it is quite different from the other group 1A elements (which is why many tables also show it in group 7A).
Predicting which atom is positive and which is negative in a covalent bond is easy if we know the electronegativities:When two atoms form a covalent bond, the atom with the lower electronegativity becomes positively charged and the atom with the higher electronegativity becomes negatively charged. (Helpful mnemonic: "The atom with a higher electronegativity becomes negative.") For example, we can use this guideline to predict the polarity of IBr. The electronegativity of I is 2.5 and that of Br is 2.8. Since 2.8 is higher than 2.5, the iodine atom is positively charged and the bromine atom is negatively charged in this molecule.

Formal charges
In general, compounds that are built entirely from nonmetals are not ionic. The atoms are held together by covalent bonds. For example, HCl is H–Cl, not H+ Cl–. The atoms may have weak electrical charges, because the bonds that link them are polar, but there are no ions present.
However, for some purposes, it is useful to carry out a sort of "electron book-keeping" and assign "make-believe" charges to the atoms in a covalently-bonded compound. For example, assigning charges to atoms can help us to predict which of two possible arrangements of atoms is more stable. For example, we can predict that the arrangement H–C–N is more stable than the arrangement H–N–C for the compound HCN. In addition, these charges help us to understand and balance oxidation-reduction reactions, in which one or more atoms change charges. (You will learn about these reactions in Chem 101B.)
The charges that we assign to atoms in a covalently-bonded molecule are called formal charges. Every atom in a molecule or polyatomic ion has a formal charge (which could be zero). For example, here are the formal charges in O3 (a molecule) and ClO3 – (a polyatomic ion):
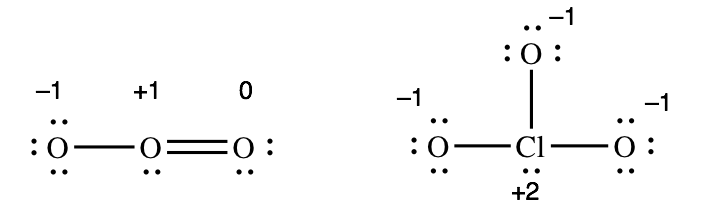
We will look at how to assign formal charges in a moment, but first, note that the formal charges must add up to the overall charge on the molecule or ion.
For O3, -1 + 1 + 0 = 0 (which matches the overall charge on O3)
For ClO3 2–, -1 + 2 + -1 + -1 = -1 (which matches the overall charge on ClO3 –)
Assigning Formal Charges
To assign formal charges to each of the atoms in any molecule or polyatomic ion, you must compare number valence electrons that each atom contributes or "brings" to the molecule to the number of electrons that the atom "owns" in the molecule.
(1) The number of electrons which an atom "brings" to the molecule is determined by its position on the periodic table. The number of electrons it "brings" is equatl to the valence electrons on the neutral atom.
(2) The number of electrons which an atom "owns" in determined based on the following:
* Non-bonding electrons "belong" to the atom on which they are located.
* Bonding electrons must be "split" between the two bond atoms involved in the bond (they are sharing after all...)
The formal charge for an atom is:
(number of electrons the atom brings to the molecule) – (number of electrons the atom actually owns in the molecule)
Here are two illustrations:
1) Formal charges on O3 (ozone):
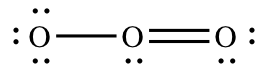
For each atom in the molecule, we must determine how many electrons it "brings" to the molecule and how many electrons it "owns" in the molecule. Oxygen is in group 6A, so each of the oxygen atoms will bring six valence electrons to the molecule. To determine the number of electrons each atom "owns" you follow the guidelines above, adding the non-bonding electrons and half of the bonding electrons. The red lines show where we must "split" the bonding electrons:
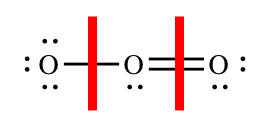
| Left oxygen atom | Center oxygen atom | Right oxygen atom | |
| number of electrons the atom brings to the molecule | 6 | 6 | 6 |
| number of electrons the atom actually owns in the molecule | 7 (six non-bonding, one bonding) | 5 (two non-bonding, three bonding) | 6 (four non-bonding, two bonding) |
| formal charge = "brings" – "owns" | -1 | +1 | 0 |
Formal charges are usually written next to the atoms in the original Lewis structure. We often omit the zeroes and just write the nonzero formal charges.
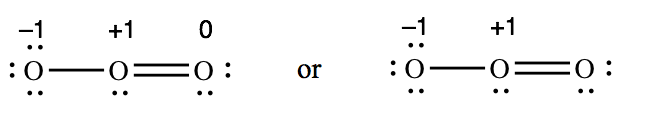
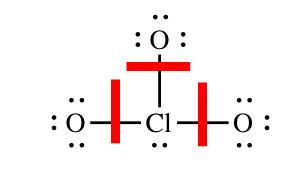
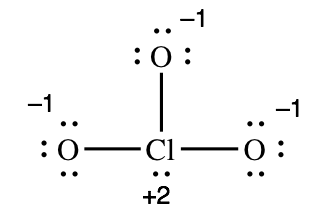
2) Formal charges on ClO3 – (chlorate ion):
For each atom in the molecule, we must determine how many electrons it "brings" to the molecule and how many electrons it "owns" in the molecule. Oxygen is in group 6A, so each of the oxygen atoms will bring six valence electrons to the molecule. Chlorine is in group 7A and bring seven valence electrons to the molecule. To determine the number of electrons each atom "owns" you follow the guidelines above, adding the non-bonding electrons and half of the bonding electrons. The red lines show where we must "split" the bonding electrons:
| Chlorine atom | Left oxygen atom | Top oxygen atom | Right oxygen atom | |
| number of electrons the atom brings to the molecule | 7 | 6 | 6 | 6 |
| number of electrons the atom actually owns in the molecule | 5 (two non-bonding, three bonding) | 7 (six non-bonding, one bonding) | 7 (six non-bonding, one bonding) | 7 (six non-bonding, one bonding) |
| formal charge = "brings" – "owns" | +2 | -1 | -1 | -1 |
Note the difference between formal charges and partial charges based on bond polarity. Formal charges are artificial charges that we calculate by breaking Lewis structures apart in a sort of electron book-keeping. We can calculate them, but they are not the true charges on the atoms. Bond polarities give us a rough idea of the true charges on atoms in a molecule, but we cannot calculate numbers. For example, the formal charges on both atoms in HCl are zero, but we know that the true charges are not zero, because the electronegativities are different.

Formal charges will prove useful tools in future sections, but for now, just focus on how to correctly calculate them.
Draw The Lewis Structure For Ibr
Source: https://chem.libretexts.org/Courses/City_College_of_San_Francisco/Chemistry_101A/Topic_F:_Molecular_Structure/09:_Basic_Concepts_of_Covalent_Bonding/9.02:_Interpreting_Lewis_Structures
Posted by: daviswallard1976.blogspot.com

0 Response to "Draw The Lewis Structure For Ibr"
Post a Comment